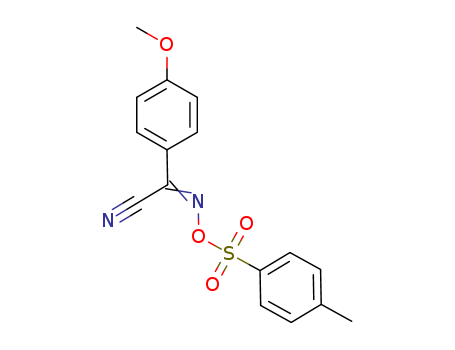
82424-53-1
- Product Name:(Z,E)-2-(4-METHOXYPHENYL)([((4-METHYLPHENYL)SULPHONYL)OXY]IMINO)ACETONITRILE
- Molecular Formula:C16H14 N2 O4 S
- Purity:99%
- Molecular Weight:330.364
Product Details
Reliable Quality (Z,E)-2-(4-METHOXYPHENYL)([((4-METHYLPHENYL)SULPHONYL)OXY]IMINO)ACETONITRILE 82424-53-1 Hot Sale with Chinese Manufacturer
- Molecular Formula:C16H14N2O4S
- Molecular Weight:330.364
- Vapor Pressure:1.17E-05mmHg at 25°C
- Boiling Point:369.7°C at 760 mmHg
- Flash Point:177.4°C
- PSA:97.13000
- Density:1.155g/cm3
- LogP:3.71758
(Z,E)-2-(4-METHOXYPHENYL)([((4-METHYLPHENYL)SULPHONYL)OXY]IMINO)ACETONITRILE(Cas 82424-53-1) Usage
|
General Description |
The chemical compound "(Z,E)-2-(4-METHOXYPHENYL)([((4-METHYLPHENYL)SULPHONYL)OXY]IMINO)ACETONITRILE" is a complex organic molecule with a long, specific name. It consists of a central acetonitrile group attached to a 4-methoxyphenyl and a 4-methylphenyl sulfonyloxyimino group. The compound can exist in two different geometric configurations, referred to as Z and E isomers. This chemical may have potential applications in organic synthesis and pharmaceutical research due to its unique structure and functional groups. Additionally, it may have biological activity or serve as a building block for the synthesis of other compounds. Further research and experimentation may be required to fully understand the properties and potential uses of this complex molecule. |
InChI:InChI=1/C14H12N2/c15-11-14(12-7-3-1-4-8-12)16-13-9-5-2-6-10-13/h1-10,14,16H/t14-/m0/s1
82424-53-1 Relevant articles
Synthesis and anti-mitotic activity of 6,7-dihydro-4H-isothiazolo[4,5-b]pyridin-5-ones: In vivo and cell-based studies
Semenov, Victor V.,Lichitsky, Boris V.,Komogortsev, Andrey N.,Dudinov, Arkady A.,Krayushkin, Mikhail M.,Konyushkin, Leonid D.,Atamanenko, Olga P.,Karmanova, Irina B.,Strelenko, Yuri A.,Shor, Boris,Semenova, Marina N.,Kiselyov, Alex S.
, p. 573 - 585 (2016/10/12)
A series of 3,7-diaryl-6,7-dihydroisothi...
82424-53-1 Process route
-
-
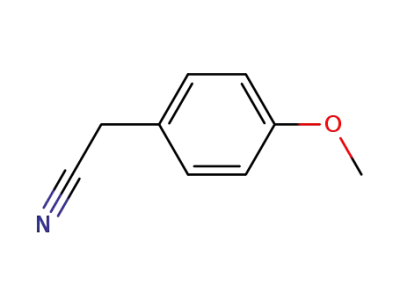
104-47-2
p-methoxybenzylnitrile

-
![-2-(4-methoxyphenyl)-2-[(4-methylphenylsulfonyl)oxyimino]acetonitrile](/upload/2025/11/fa2546e6-8341-46be-ba34-b269371ac656.png)
-
82424-53-1
-2-(4-methoxyphenyl)-2-[(4-methylphenylsulfonyl)oxyimino]acetonitrile
| Conditions | Yield |
|---|---|
|
Multi-step reaction with 2 steps
1: isopentyl nitrite; sodium methylate / methanol / 1 h / 5 °C
2: N,N-dimethyl-formamide / 0.25 h / 75 °C
With
sodium methylate; isopentyl nitrite;
In
methanol; N,N-dimethyl-formamide;
|
-
-
98-59-9
p-toluenesulfonyl chloride

-
-
C9H7N2O2(1-)*Na(1+)

-
![-2-(4-methoxyphenyl)-2-[(4-methylphenylsulfonyl)oxyimino]acetonitrile](/upload/2025/11/fa2546e6-8341-46be-ba34-b269371ac656.png)
-
82424-53-1
-2-(4-methoxyphenyl)-2-[(4-methylphenylsulfonyl)oxyimino]acetonitrile
| Conditions | Yield |
|---|---|
|
In
N,N-dimethyl-formamide;
at 75 ℃;
for 0.25h;
|
82424-53-1 Upstream products
-
98-59-9

p-toluenesulfonyl chloride
-
104-47-2

p-methoxybenzylnitrile
Relevant Products
-
1-[4-(Phenylthio)phenyl]-1,2-octanedione 2-(O-benzoyloxime)
CAS:253585-83-0
-
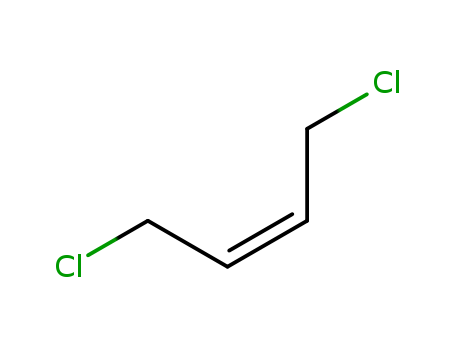
cis-1,4-Dichloro-2-butene
CAS:1476-11-5