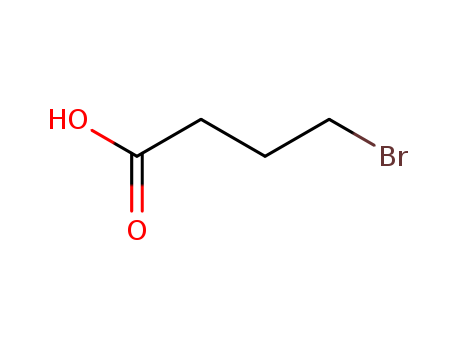
2623-87-2
- Product Name:4-Bromobutyric acid
- Molecular Formula:C4H7BrO2
- Purity:98% GC
- Molecular Weight:167.002
Product Details
Quality Factory Hot Selling 4-Bromobutyric acid 2623-87-2 with Fast Shipping
- Molecular Formula:C4H7BrO2
- Molecular Weight:167.002
- Appearance/Colour:slightly yellow solid
- Vapor Pressure:0.00763mmHg at 25°C
- Melting Point:31 - 33 °C
- Refractive Index:1.4730 (estimate)
- Boiling Point:248.7 °C at 760 mmHg
- PKA:4.51±0.10(Predicted)
- Flash Point:111.3 °C
- PSA:37.30000
- Density:1.631 g/cm3
- LogP:1.24610
4-Bromobutyric acid(Cas 2623-87-2) Usage
|
Synthesis |
4-Bromobutyric acid may be produced by a reaction of Y-butyrolactone with hydrogen bromide. The hydrogen bromide to be used may be gas or an aqueous Solution thereof. The reaction temperature is usually about 10 to 100°C., and the amount of hydrogen bromide to be used is usually about 1 to 10 moles per mole of Y-butyrolactone. |
InChI:InChI=1/C4H7BrO2/c5-3-1-2-4(6)7/h1-3H2,(H,6,7)/p-1
2623-87-2 Relevant articles
Synthesis and Properties of Novel Surface Active Monomers Based on Derivatives of 4-Hydroxybutyric Acid and 6-Hydroxyhexanoic Acid
Borzenkov, Mykola,Mitina, Natalia,Lobaz, Volodymyr,Hevus, Orest
, p. 133 - 144 (2015)
Novel surface active maleate and methacr...
-
Raevskii,K.S. et al.
, (1976)
-
Boron tribromide as a reagent for anti-Markovnikov addition of HBr to cyclopropanes
Chen, Shuming,Gieuw, Matthew H.,Houk, K. N.,Ke, Zhihai,Yeung, Ying-Yeung
, p. 9426 - 9433 (2020/10/02)
Although radical formation from a trialk...
Synthesis of polysiloxane-based quaternized imidazolium salts with a hydroxy group at the end of alkyl groups
Ichikawa, Tsukasa,Wako, Tsuyoshi,Nemoto, Nobukatsu
, p. 1 - 8 (2015/12/18)
A series of polysiloxane derivatives hav...
Building blocks for (C15-C3)-modified epothilone D analogs
Valeev,Bikzhanov,Miftakhov
, p. 1511 - 1519 (2015/02/02)
A promising potentially biologically act...
2623-87-2 Process route
-
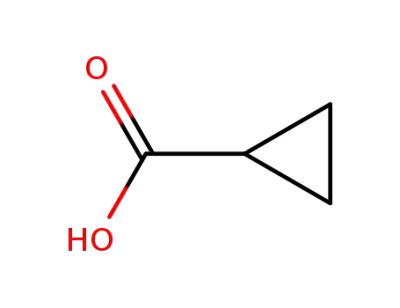
-
1759-53-1
cyclopropanecarboxylic acid

-
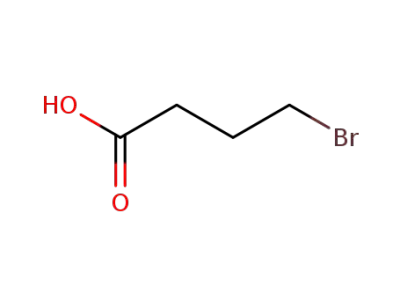
-
2623-87-2
bromobutyric acid
| Conditions | Yield |
|---|---|
|
With
water; boron tribromide;
In
dichloromethane;
at 23 ℃;
for 46h;
regioselective reaction;
|
67% |
|
With
hydrogen bromide;
at 175 ℃;
|
-
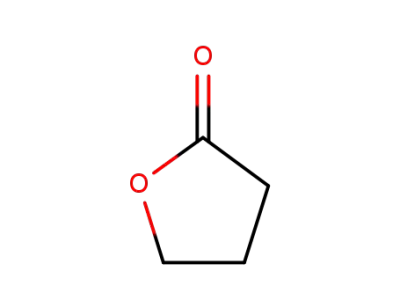
-
96-48-0
4-butanolide

-

-
2623-87-2
bromobutyric acid
| Conditions | Yield |
|---|---|
|
With
tetrabutylammomium bromide; hydrogen bromide;
In
water;
for 0.166667h;
Microwave irradiation;
|
87% |
|
With
trimethylsilyl bromide;
iodine(I) bromide;
for 12h;
Ambient temperature;
|
73% |
|
With
sulfuric acid; hydrogen bromide;
In
water;
for 12h;
Inert atmosphere;
Reflux;
|
65% |
|
With
sulfuric acid; hydrogen bromide;
at 20 ℃;
for 7h;
Reflux;
|
61% |
|
With
hydrogen bromide;
at 120 - 130 ℃;
for 24h;
Heating;
|
36.4% |
|
With
hydrogen bromide;
|
|
|
With
hydrogen bromide;
|
|
|
With
water; boron tribromide;
Yield given. Multistep reaction;
1.) dichloromethane, room temp., 16 h, 2.) 0 deg C;
|
|
|
With
hydrogen bromide; acetic acid;
at 20 - 70 ℃;
for 4h;
|
|
|
With
hydrogen bromide;
In
water;
|
|
|
With
sulfuric acid; hydrogen bromide;
|
2623-87-2 Upstream products
-
6303-58-8
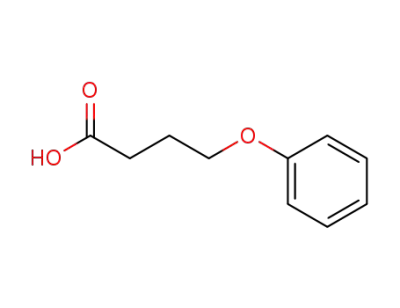
4-phenyloxybutanoic acid
-
625-38-7
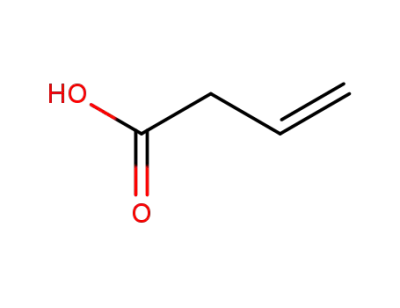
but-3-enoic acid
-
10374-37-5
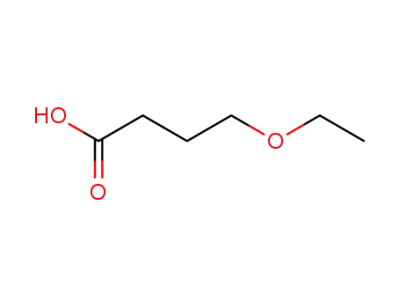
4-ethoxybutanoic acid
-
28341-54-0
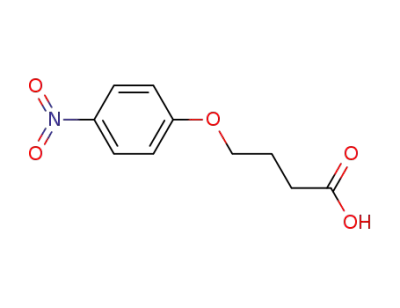
4-(4-nitrophenoxy)butanoic acid
2623-87-2 Downstream products
-
4897-84-1
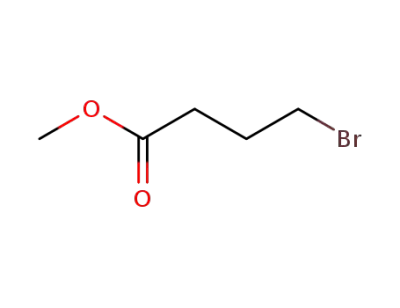
Methyl 4-bromobutyrate
-
2969-81-5
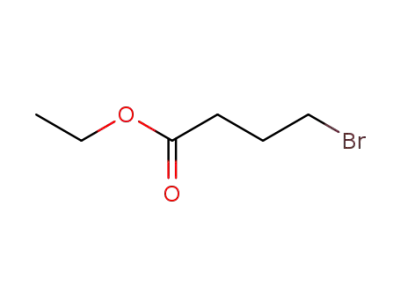
Ethyl 4-bromobutyrate
-
30845-11-5
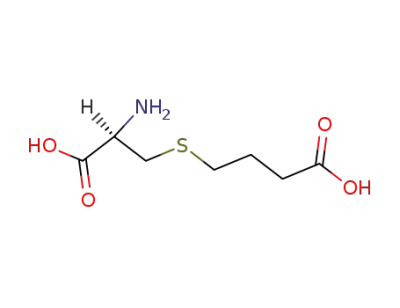
S-(3-Carboxypropyl)-cystein
-
96-48-0

4-butanolide
Relevant Products
-
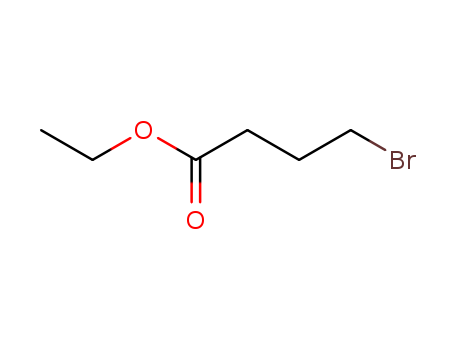
Ethyl 4-bromobutyrate
CAS:2969-81-5
-
2-Bromoacetamide
CAS:683-57-8
-
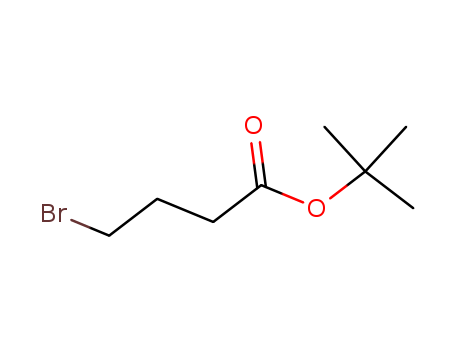
tert-Butyl 4-bromobutanoate
CAS:110661-91-1