75726-96-4
- Product Name:2-bromo-N-isopropylacetamide
- Molecular Formula:C5H10BrNO
- Purity:98% GC
- Molecular Weight:180.044
Product Details
Manufacturer Supply Best Quality 2-bromo-N-isopropylacetamide 75726-96-4 with Efficient Transportation
- Molecular Formula:C5H10BrNO
- Molecular Weight:180.044
- Vapor Pressure:0.009mmHg at 25°C
- Melting Point:63-64℃
- Refractive Index:1.472
- Boiling Point:265.834 °C at 760 mmHg
- PKA:13.71±0.46(Predicted)
- Flash Point:114.573 °C
- PSA:29.10000
- Density:1.379 g/cm3
- LogP:1.29680
2-Bromo-N-isopropylacetamide(Cas 75726-96-4) Usage
|
General Description |
2-Bromo-N-isopropylacetamide is a chemical compound that belongs to the class of amides and isopropyl compounds. It is typically used as an intermediate in the synthesis of various pharmaceuticals and organic compounds. The presence of the bromo group makes this compound a valuable building block in organic synthesis, allowing for the introduction of diverse functional groups through substitution reactions. Additionally, the isopropyl group provides steric hindrance, which can influence the reactivity and selectivity of certain reactions. Overall, 2-Bromo-N-isopropylacetamide plays a crucial role in the production of various pharmaceutical and organic products due to its versatile chemical properties. |
InChI:InChI=1/C5H10BrNO/c1-4(2)7-5(8)3-6/h4H,3H2,1-2H3,(H,7,8)
75726-96-4 Relevant articles
In Vitro Anti-Toxoplasma gondii Activity Evaluation of a New Series of Quinazolin-4(3H)-one Derivatives
Deng, Yu,Mu, Hao,Li, Hong-Bo,Fu, Li-Zhi,Tang, Da,Wu, Tao,Huang, Shu-Heng,Li, Cheng-Hong
, (2021/11/18)
Toxoplasmosis post serious threaten to h...
Synthesis method of SLx-2119
-
Paragraph 0017; 0035-0038; 0058; 0066, (2017/08/30)
The invention belongs to the field of dr...
TREATMENT OF GVHD
-
Paragraph 0192, (2015/11/18)
The invention relates to treatment of gr...
AUTOTAXIN INHIBITOR COMPOUNDS
-
Paragraph 00432, (2015/04/15)
Described herein are compounds that are ...
75726-96-4 Process route
-
-
75-31-0
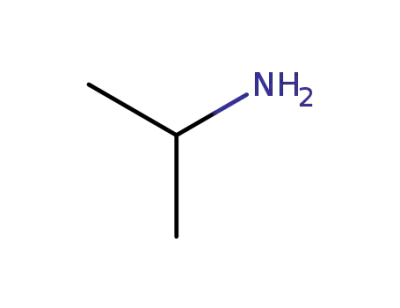
isopropylamine

-
-
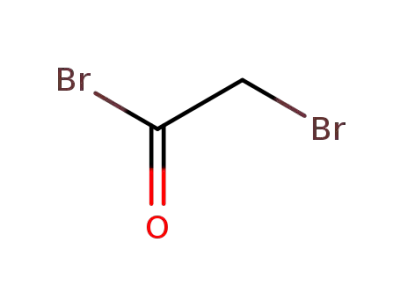
598-21-0
2-Bromoacetyl bromide

-
-
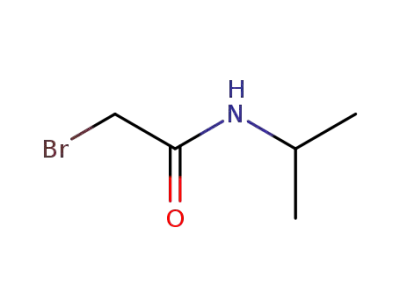
75726-96-4
2-bromo-N-isopropylacetamide
| Conditions | Yield |
|---|---|
|
In
dichloromethane;
at 0 - 20 ℃;
|
85% |
|
In
dichloromethane;
at 20 ℃;
for 1h;
Concentration;
Inert atmosphere;
Cooling with ice;
|
71% |
|
In
1,2-dichloro-ethane;
at -10 ℃;
for 0.166667h;
|
70% |
|
In
1,2-dichloro-ethane;
at -10 ℃;
for 0.166667h;
|
70% |
|
In
dichloromethane;
at -10 ℃;
for 0.166667h;
|
70% |
|
In
dichloromethane;
at -10 ℃;
for 0.166667h;
|
70% |
|
In
1,2-dichloro-ethane;
at -10 ℃;
for 0.166667h;
|
70% |
|
With
potassium carbonate;
In
dichloromethane;
at 0 - 20 ℃;
|
41% |
|
In
dichloromethane;
at 20 ℃;
for 0.583333h;
Inert atmosphere;
|
31% |
|
With
1,2-dichloro-ethane;
at -10 ℃;
|
|
|
With
sodium hydrogencarbonate;
In
tetrahydrofuran;
at -78 - 20 ℃;
for 14h;
|
|
|
With
triethylamine;
In
dichloromethane;
Inert atmosphere;
Cooling with ice;
|
-
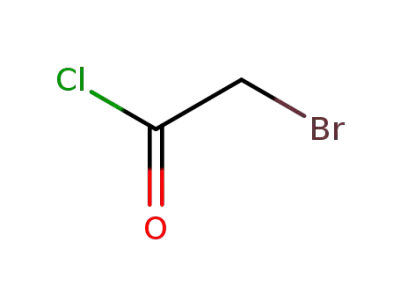
-
22118-09-8
2-bromoacetyl chloride

-

-
75-31-0
isopropylamine

-

-
75726-96-4
2-bromo-N-isopropylacetamide
| Conditions | Yield |
|---|---|
|
With
triethylamine;
In
chloroform;
at -50 - -15 ℃;
|
44% |
|
With
4-methyl-morpholine;
In
dichloromethane;
at -15 ℃;
|
|
|
|
|
|
With
triethylamine;
In
N,N-dimethyl-formamide;
at 0 - 20 ℃;
for 2.5h;
Inert atmosphere;
|
|
|
In
N,N-dimethyl-formamide;
|
75726-96-4 Upstream products
-
75-31-0

isopropylamine
-
598-21-0

2-Bromoacetyl bromide
-
71985-62-1
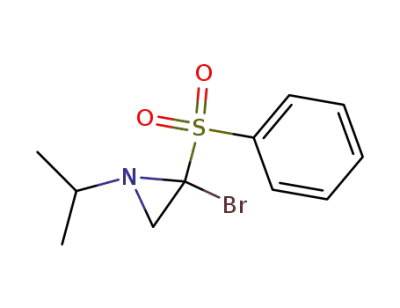
2-benzenesulfonyl-2-bromo-1-isopropyl-aziridine
-
22118-09-8

2-bromoacetyl chloride
75726-96-4 Downstream products
-
38265-81-5
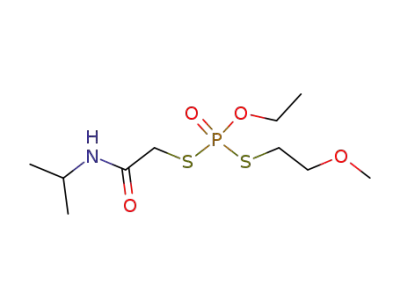
Dithiophosphoric acid O-ethyl ester S-(isopropylcarbamoyl-methyl) ester S'-(2-methoxy-ethyl) ester
-
38265-82-6
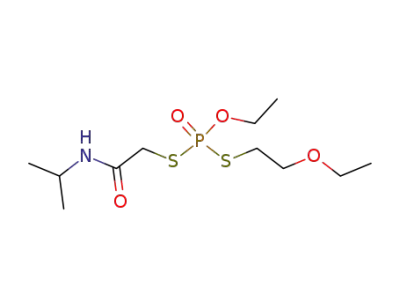
Dithiophosphoric acid S-(2-ethoxy-ethyl) ester O-ethyl ester S'-(isopropylcarbamoyl-methyl) ester
-
151965-03-6
2-Benzyloxyamino-N-isopropyl-acetamide
-
104241-10-3
C59H91N18O22S3(1+)*HO4S(1-)
Relevant Products
-
2-Bromoacetamide
CAS:683-57-8
-
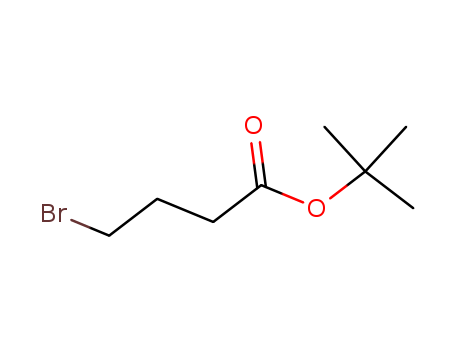
tert-Butyl 4-bromobutanoate
CAS:110661-91-1
-
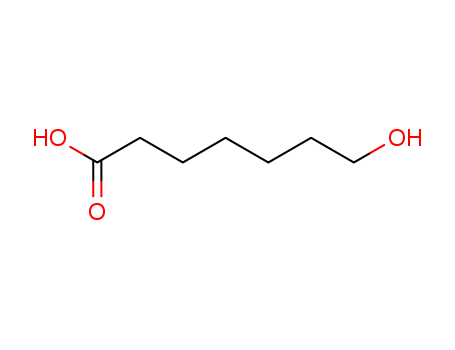
7-Hydroxyheptanoic Acid
CAS:3710-42-7