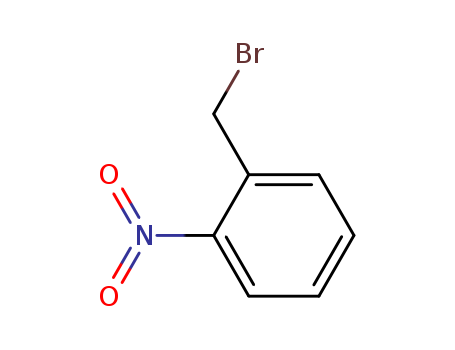
3958-60-9
- Product Name:2-Nitrobenzyl bromide
- Molecular Formula:C7H6BrNO2
- Purity:98% GC
- Molecular Weight:216.034
Product Details
Excellent chemical plant bulk supply 2-Nitrobenzyl bromide 3958-60-9
- Molecular Formula:C7H6BrNO2
- Molecular Weight:216.034
- Appearance/Colour:white to light yellow crystal powder
- Vapor Pressure:0.00868mmHg at 25°C
- Melting Point:44-46 °C(lit.)
- Refractive Index:1.611
- Boiling Point:275.2 °C at 760 mmHg
- Flash Point:120.2 °C
- PSA:45.82000
- Density:1.652 g/cm3
- LogP:3.01290
2-Nitrobenzyl bromide(Cas 3958-60-9) Usage
|
Biochem/physiol Actions |
2-Nitrobenzyl bromide reacts with L-cysteine to form S-2-nitrobenzyl-cysteine which was used for modification of ultra-low-gelling-temperature (ULGT) agarose. |
InChI:InChI=1/C7H6BrNO2/c8-5-6-3-1-2-4-7(6)9(10)11/h1-4H,5H2
3958-60-9 Relevant articles
A photo-degradable injectable self-healing hydrogel based on star poly(ethylene glycol)-: B -polypeptide as a potential pharmaceuticals delivery carrier
Zhao, Dinglei,Tang, Quan,Zhou, Qiang,Peng, Kang,Yang, Haiyang,Zhang, Xingyuan
, p. 7420 - 7428 (2018)
As one of the most promising biomaterial...
Design, synthesis, biological screening and molecular docking studies of novel multifunctional 1,4-di (aryl/heteroaryl) substituted piperazine derivatives as potential antitubercular and antimicrobial agents
Mekonnen Sanka, Bruktawit,Mamo Tadesse, Dereje,Teju Bedada, Endale,Mengesha, Ephriem T.,Babu G., Neelaiah
, (2022/01/20)
In this paper, two series of novel multi...
Ethylene homo- and copolymerization catalyzed by vanadium, zirconium, and titanium complexes having potentially tridentate Schiff base ligands
Bia?ek, Marzena,Fryga, Julia,Hajdasz, Natalia,Matsko, Mikhail A.,Spaleniak, Grzegorz
, p. 184 - 194 (2021/07/09)
New potentially tridentate Schiff base l...
NovelN-transfer reagent for converting α-amino acid derivatives to α-diazo compounds
Lu, Guan-Han,Huang, Tzu-Chia,Hsueh, Hsiao-Chin,Yang, Shin-Cherng,Cho, Ting-Wei,Chou, Ho-Hsuan
supporting information, p. 4839 - 4842 (2021/05/25)
A novel universalN-transfer reagent for ...
N-transfer reagent and method for preparing the same and its application
-
Page/Page column 16-17, (2021/06/25)
Provided are a novel N-transfer reagent ...
3958-60-9 Process route
-
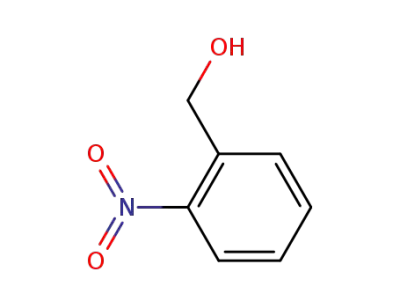
-
612-25-9
2-Nitrobenzyl alcohol

-
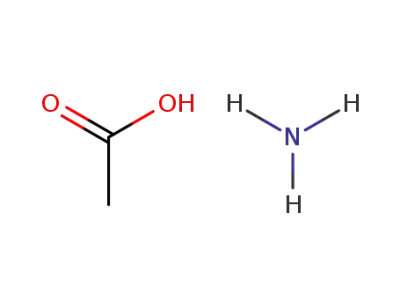
-
631-61-8,92206-38-7
ammonium acetate

-
-
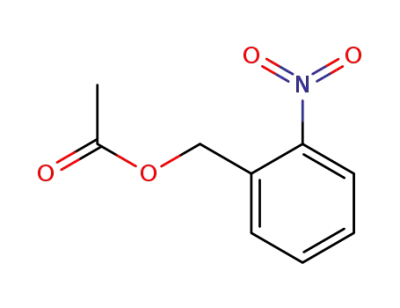
77376-01-3
acetic acid 2-nitro-benzyl ester

-
-
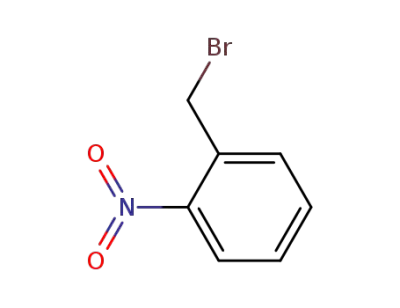
3958-60-9
2-nitrophenylmethyl bromide
| Conditions | Yield |
|---|---|
|
With
N-Bromosuccinimide; triphenylphosphine;
In
acetonitrile;
at 20 ℃;
for 12h;
Cooling with ice;
|
78% 15% |
-
-
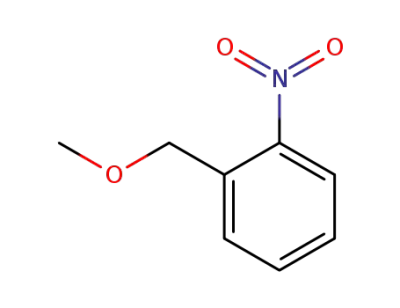
38177-30-9
1-(methoxymethyl)-2-nitrobenzene

-

-
3958-60-9
2-nitrophenylmethyl bromide
| Conditions | Yield |
|---|---|
|
With
hydrogen bromide; acetic acid;
at 65 ℃;
for 2h;
Inert atmosphere;
|
95.1% |
3958-60-9 Upstream products
-
56-23-5
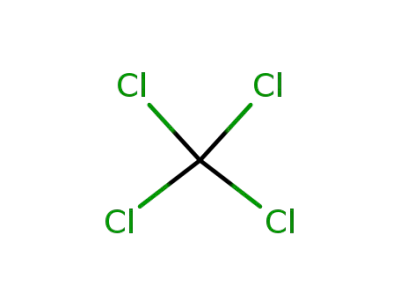
tetrachloromethane
-
128-08-5
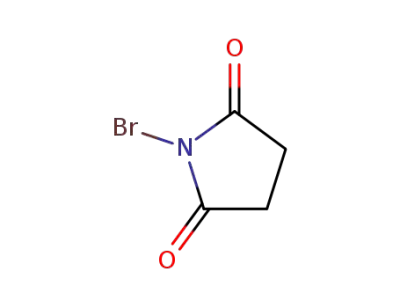
N-Bromosuccinimide
-
88-72-2
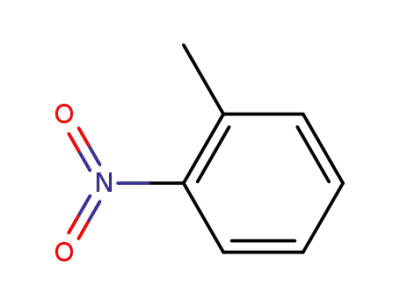
1-methyl-2-nitrobenzene
-
94-36-0
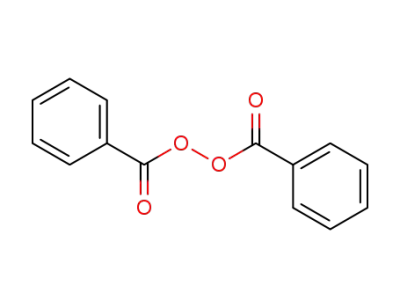
dibenzoyl peroxide
3958-60-9 Downstream products
-
13664-80-7
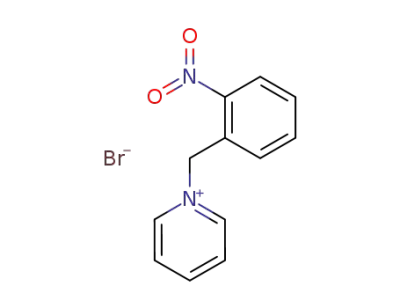
1-(2-nitro-benzyl)-pyridinium; bromide
-
109407-56-9

1-(2-nitro-benzyloxy)-pyridinium; bromide
-
1154-99-0
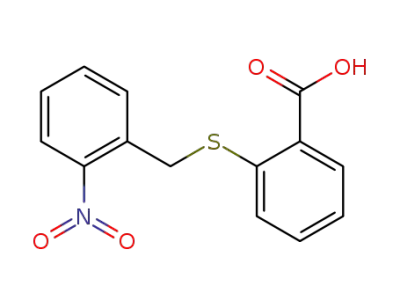
2-(2-nitro-benzylsulfanyl)-benzoic acid
-
55581-64-1
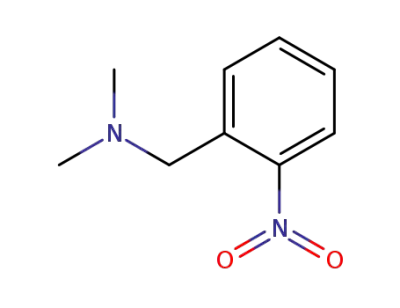
N,N-dimethyl-1-(2-nitrophenyl)methylamine
Relevant Products
-
2-Bromoacetamide
CAS:683-57-8
-
7-Hydroxyheptanoic Acid
CAS:3710-42-7
-
7-Bromoheptanoic acid
CAS:30515-28-7