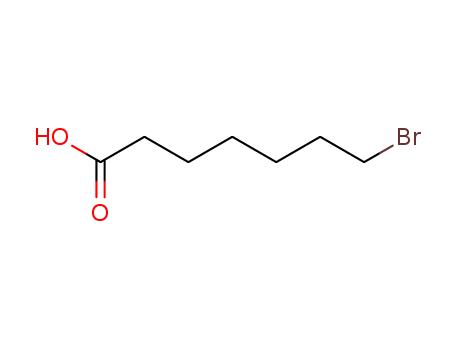
30515-28-7
- Product Name:7-Bromoheptanoic acid
- Molecular Formula:C7H13BrO2
- Purity:97% GC
- Molecular Weight:209.083
Product Details
Factory Supply industrial standard 7-Bromoheptanoic acid 30515-28-7 In Stock
- Molecular Formula:C7H13BrO2
- Molecular Weight:209.083
- Vapor Pressure:0.000985mmHg at 25°C
- Melting Point:136-138 °C
- Refractive Index:1.488
- Boiling Point:280.7 °C at 760 mmHg
- PKA:4.76±0.10(Predicted)
- Flash Point:123.6 °C
- PSA:37.30000
- Density:1.376 g/cm3
- LogP:2.41640
7-Bromoheptanoic acid(Cas 30515-28-7) Usage
InChI:InChI=1/C7H13BrO2/c8-6-4-2-1-3-5-7(9)10/h1-6H2,(H,9,10)
30515-28-7 Relevant articles
-
Smith,J.R.L. et al.
, p. 1381 - 1383 (1978)
-
Employing in vitro metabolism to guide design of F-labelled PET probes of novel α-synuclein binding bifunctional compounds
Aigbogun, Omozojie P.,Allen, Kevin J. H,Krol, Ed S.,Lee, Jeremy S.,Nwabufo, Chukwunonso K.,Owens, Madeline N.,Phenix, Christopher P.
supporting information, p. 885 - 900 (2021/07/09)
A challenge in the development of novel ...
Illiberis ulmivora Graeser sex attractant and preparation method thereof
-
, (2021/07/01)
The invention provides an Illiberis ulmi...
Hydroxamic acid derivative and JHDM inhibitor
-
Paragraph 0053, (2016/10/09)
PROBLEM TO BE SOLVED: To provide a compo...
Total Syntheses of (R)-Strongylodiols C and D
Liu, Feipeng,Zhong, Jiangchun,Li, Shuoning,Li, Minyan,Wu, Lin,Wang, Qian,Mao, Jianyou,Liu, Shikuo,Zheng, Bing,Wang, Min,Bian, Qinghua
supporting information, p. 244 - 247 (2016/02/05)
The first total syntheses of two marine ...
30515-28-7 Process route
-
-
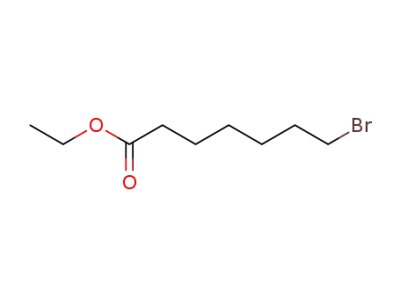
29823-18-5
ethyl 7-bromoheptanoate

-
-
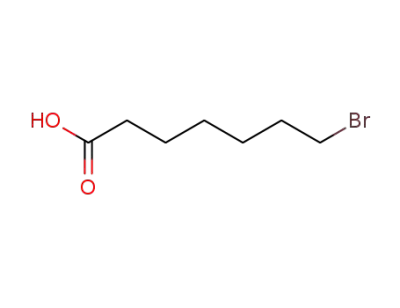
30515-28-7
7-bromoheptanoic acid
| Conditions | Yield |
|---|---|
|
With
lithium hydroxide;
In
tetrahydrofuran; ethanol;
at 20 ℃;
|
100% |
|
With
lithium hydroxide;
In
ethanol;
at 20 ℃;
|
99% |
|
With
lithium hydroxide;
In
tetrahydrofuran; ethanol;
at 20 ℃;
|
99% |
|
With
lithium hydroxide;
In
tetrahydrofuran; ethanol; water;
at 20 ℃;
|
99% |
|
With
lithium hydroxide;
In
tetrahydrofuran; ethanol; water;
at 20 ℃;
for 3h;
|
98% |
|
ethyl 7-bromoheptanoate;
With
water; lithium hydroxide;
In
ethanol;
at 20 ℃;
With
hydrogenchloride;
In
ethanol; water;
Cooling with ice;
|
96% |
|
With
lithium hydroxide;
In
methanol;
at 20 ℃;
for 1h;
|
89% |
|
With
water; hydrogen bromide;
at 130 ℃;
for 5h;
|
86% |
|
With
lithium hydroxide monohydrate;
In
tetrahydrofuran; ethanol; water;
at 20 ℃;
for 7h;
|
86% |
|
With
hydrogen bromide;
for 5h;
Heating;
|
80% |
|
With
lithium hydroxide;
In
tetrahydrofuran; methanol;
for 4h;
Heating;
|
66% |
|
With
water; lithium hydroxide;
In
methanol;
at 20 ℃;
for 0.5h;
|
-
-
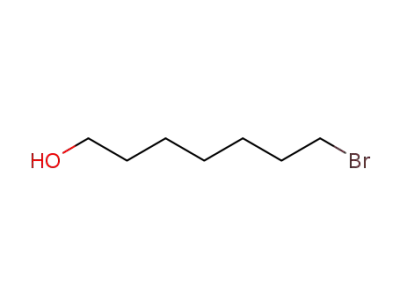
10160-24-4
7-bromoheptyl alcohol

-

-
30515-28-7
7-bromoheptanoic acid
| Conditions | Yield |
|---|---|
|
With
nitric acid;
at 20 - 60 ℃;
for 5.75h;
Inert atmosphere;
|
95% |
|
With
nitric acid;
at 20 - 60 ℃;
for 4.75h;
|
95% |
|
With
nitric acid;
at 20 - 60 ℃;
for 4h;
|
95% |
|
7-bromoheptyl alcohol;
With
sodium hypochlorite; 2,2,6,6-Tetramethyl-1-piperidinyloxy free radical; tetrabutylammomium bromide; sodium hydrogencarbonate;
In
water; ethyl acetate;
at 0 ℃;
With
hydrogenchloride;
In
water;
|
84% |
|
With
chromium(VI) oxide;
In
acetic acid;
for 20h;
Ambient temperature;
|
30515-28-7 Upstream products
-
20965-27-9

7-bromo-heptanonitrile
-
20395-28-2
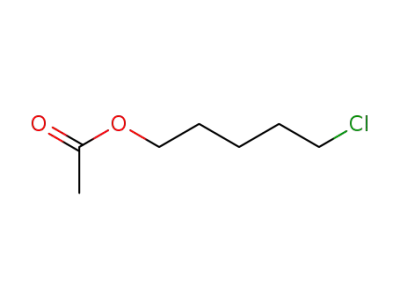
5-chloropentyl acetate
-
996-82-7

sodium diethylmalonate
-
1906-95-2
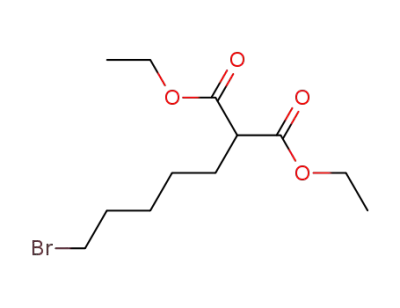
5-bromopentylmalonic acid diethyl ester
30515-28-7 Downstream products
-
29823-18-5

ethyl 7-bromoheptanoate
-
123-99-9

azelaic acid
-
3344-70-5

1,12-dibromododecane
-
50733-91-0
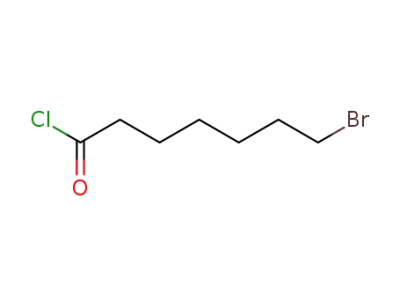
7-bromoheptanoyl chloride
Relevant Products
-
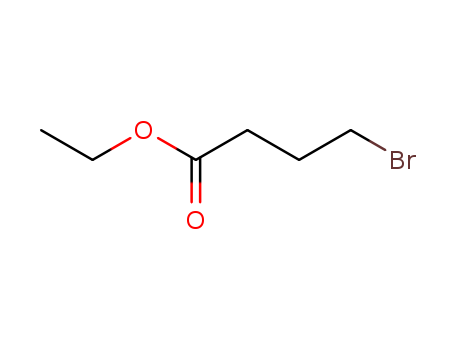
Ethyl 4-bromobutyrate
CAS:2969-81-5
-
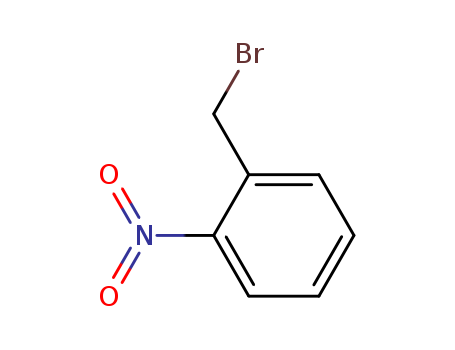
2-Nitrobenzyl bromide
CAS:3958-60-9
-
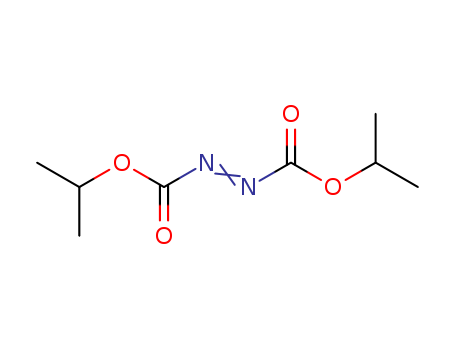
Diisopropyl azodicarboxylate
CAS:2446-83-5