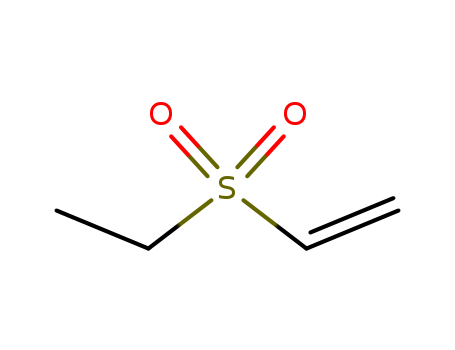
1889-59-4
- Product Name:Ethyl vinyl sulfone
- Molecular Formula:C4H8O2S
- Purity:97% GC
- Molecular Weight:120.172
Product Details
Top Quality Ethyl vinyl sulfone 1889-59-4 Hot Sell In Stock
- Molecular Formula:C4H8O2S
- Molecular Weight:120.172
- Appearance/Colour:clear yellow liquid
- Vapor Pressure:0.0705mmHg at 25°C
- Refractive Index:n20/D 1.463(lit.)
- Boiling Point:237 °C at 760 mmHg
- Flash Point:109 °C
- PSA:42.52000
- Density:1.085 g/cm3
- LogP:1.64540
Ethyl vinyl sulfone(Cas 1889-59-4) Usage
|
General Description |
Ethly vinyl sulfone alkylates ε-amino groups of lysine side chains and imidazole groups of histidine residues in proteins. Chemical modification of bovine serum albumin by ethyl vinyl sulfone has been studied by X-ray photoelectron spectroscopy. |
InChI:InChI=1/C4H8O2S/c1-3-7(5,6)4-2/h3H,1,4H2,2H3
1889-59-4 Relevant articles
-
Sayigh,A.A.R. et al.
, p. 2042 - 2043 (1964)
-
Rates of Reaction of the Sulfoxides and Sulfones of Sulfur Mustard and 2-Chloroethyl Ethyl Sulfide with Hydroxide Ion in Water
Tilley, R. Ian,Leslie, D. Ralph
, p. 1781 - 1786 (1995)
The reactions of the sulfoxides and sulf...
Mo(VI) complex catalysed synthesis of sulfonees and their modification for anti-HIV activities
Madduluri, Vimal Kumar,Baig, Noorullah,Chander, Subhash,Murugesan, Sankaranarayanan,Sah, Ajay K.
, (2020/01/23)
An efficient method for the synthesis of...
A safe and compact flow platform for the neutralization of a mustard gas simulant with air and light
Bianchi, Pauline,Emmanuel, Noémie,Legros, Julien,Monbaliu, Jean-Christophe M.
supporting information, p. 4105 - 4115 (2020/07/30)
A low footprint, mobile, robust and frug...
Difluoro- and trifluoro diazoalkanes-complementary approaches in batch and flow and their application in cycloaddition reactions
Hock, Katharina J.,Mertens, Lucas,Metze, Friederike K.,Schmittmann, Clemens,Koenigs, Rene M.
supporting information, p. 905 - 909 (2017/08/14)
Herein we report on applications of fluo...
Flash vacuum pyrolysis of stabilised phosphorus ylides. Part 15. Generation of alkoxycarbonyl(sulfenyl)carbenes and their intramolecular insertion to give alkenyl sulfides
Aitken, R. Alan,Armstrong, Jill M.,Drysdale, Martin J.,Ross, Fiona C.,Ryan, Bruce M.
, p. 593 - 604 (2007/10/03)
A range of 18 alkoxycarbonyl sulfinyl ph...
1889-59-4 Process route
-
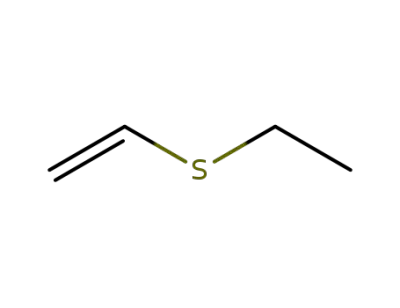
-
627-50-9
vinyl ethylsulfide

-
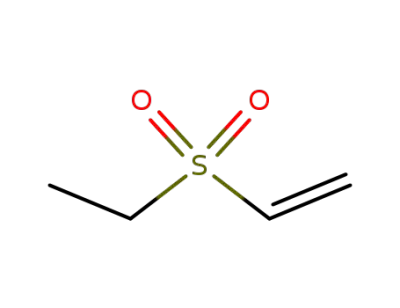
-
1889-59-4
ethyl vinyl sulfone
| Conditions | Yield |
|---|---|
|
With
urea hydrogen peroxide adduct;
In
ethanol;
at 20 ℃;
for 0.25h;
|
95% |
|
With
monoperoxyphthalic acid; diethyl ether;
|
|
|
With
pyridine; dihydrogen peroxide;
at 70 - 80 ℃;
|
|
|
With
oxone;
In
methanol; water;
at 20 ℃;
|
|
|
With
pyridine; dihydrogen peroxide;
at 70 - 80 ℃;
|
-
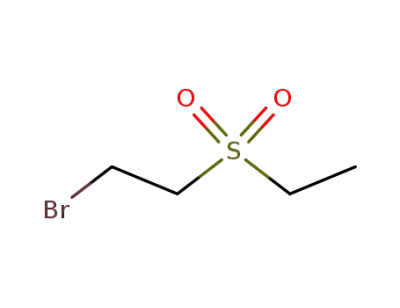
-
30129-83-0
1-bromo-2-ethanesulfonyl-ethane

-

-
1889-59-4
ethyl vinyl sulfone
| Conditions | Yield |
|---|---|
|
With
potassium hydroxide;
at 25 ℃;
|
|
|
With
trimethylamine; benzene;
|
1889-59-4 Upstream products
-
25027-40-1
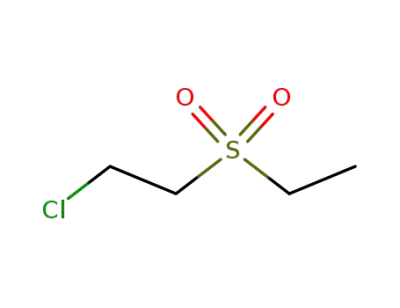
2-chloroethyl ethyl sulfone
-
30129-83-0

1-bromo-2-ethanesulfonyl-ethane
-
513-12-2
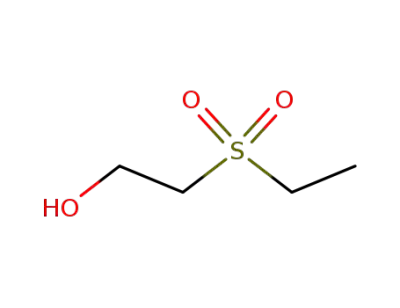
2-ethanesulfonylethanol
-
121-44-8

triethylamine
1889-59-4 Downstream products
-
72034-97-0
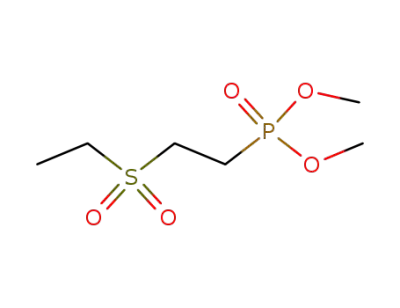
(2-ethanesulfonyl-ethyl)-phosphonic acid dimethyl ester
-
98560-85-1
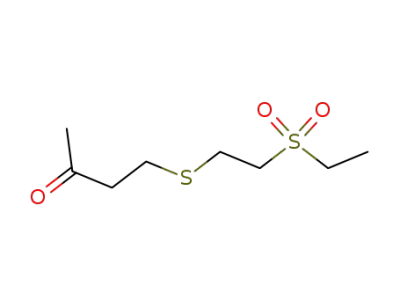
4-(2-ethanesulfonyl-ethylsulfanyl)-butan-2-one
-
513-12-2

2-ethanesulfonylethanol
-
66754-40-3
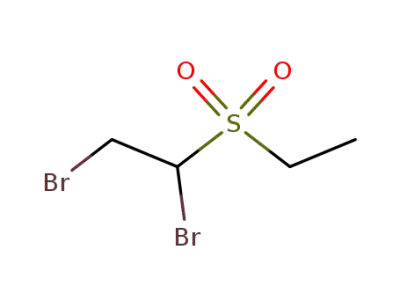
1,2-dibromo-1-ethanesulfonyl-ethane
Relevant Products
-
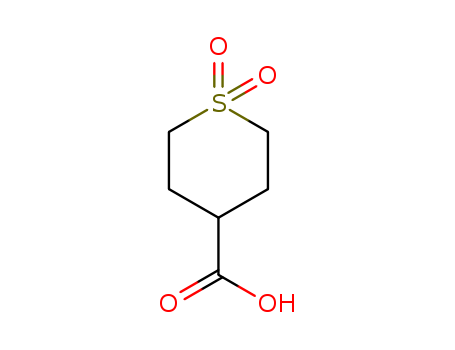
1,1-Dioxo-tetrahydrothiopyran-4-carboxylic acid
CAS:64096-87-3
-
1-[9-(2-Ethylhexyl)-6-nitro-9H-carbazol-3-yl]ethanone O-acetyloxime
CAS:1035539-18-4
-
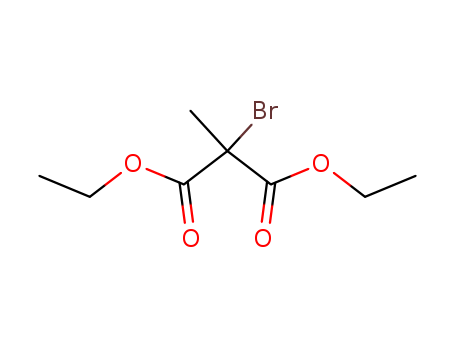
Diethyl 2-bromo-2-methylmalonate
CAS:29263-94-3