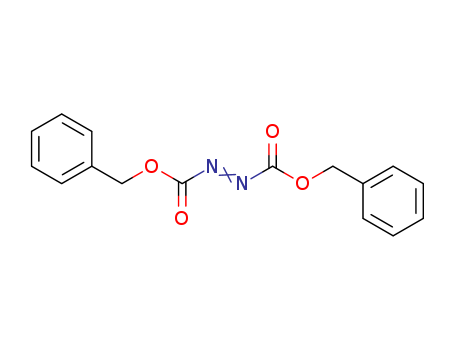
2449-05-0
- Product Name:Dibenzyl azodicarboxylate
- Molecular Formula:C16H14N2O4
- Purity:98% HPLC
- Molecular Weight:298.298
Product Details
Factory sells Dibenzyl azodicarboxylate 2449-05-0 with sufficient production capacity
- Molecular Formula:C16H14N2O4
- Molecular Weight:298.298
- Appearance/Colour:Yellow to orange crystalline powder
- Vapor Pressure:1.57E-08mmHg at 25°C
- Melting Point:43-47 °C(lit.)
- Refractive Index:1.567
- Boiling Point:456.8 °C at 760 mmHg
- Flash Point:203 °C
- PSA:77.32000
- Density:1.19 g/cm3
- LogP:4.11220
Dibenzyl azodicarboxylate(Cas 2449-05-0) Usage
|
General Description |
Dibenzyl azodicarboxylate undergoes[4+2] cycloaddition reaction with glycal to yield 2-aminoglycosides. |
InChI:InChI=1/C16H14N2O4/c19-15(21-11-13-7-3-1-4-8-13)17-18-16(20)22-12-14-9-5-2-6-10-14/h1-10H,11-12H2/b18-17+
2449-05-0 Relevant articles
Synthesis of α-aminocarbonyl compounds via hetero dielsalder reaction
Sakurai, Masayoshi,Kihara, Nobuhiro,Watanabe, Nobuhiro,Ikari, Yoshihiro,Takata, Toshikazu
supporting information, p. 144 - 147 (2018/01/01)
A synthetic route to α-aminoketone deriv...
Photocatalytic esterification under Mitsunobu reaction conditions mediated by flavin and visible light
M?rz,Chudoba,Kohout,Cibulka
supporting information, p. 1970 - 1975 (2017/03/11)
The usefulness of flavin-based aerial ph...
Metal-free oxyaminations of alkenes using hydroxamic acids
Schmidt, Valerie A.,Alexanian, Erik J.
supporting information; scheme or table, p. 11402 - 11405 (2011/09/16)
A radical-mediated approach to metal-fre...
An improved oxidation method for the synthesis of azodicarbonyl compounds
Starr, Jeremy T.,Rai, Gurdev S.,Dang, Hung,McNelis, Brian J.
, p. 3197 - 3200 (2007/10/03)
Five representative hydrazo compounds ha...
2449-05-0 Process route
-
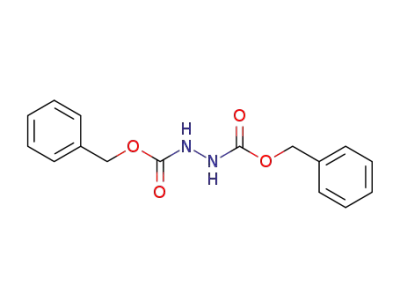
-
5394-50-3
bis(phenylmethyl) 1,2-hydrazinedicarboxylate

-
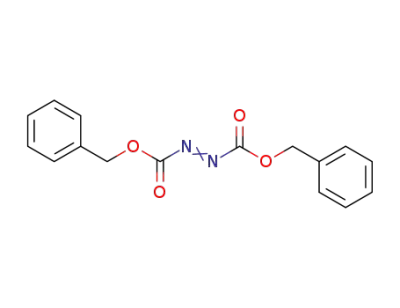
-
2449-05-0
dibenzyl azodicarboxylate
| Conditions | Yield |
|---|---|
|
With
pyridine; bromine;
In
dichloromethane;
at 0 ℃;
for 1.5h;
Inert atmosphere;
|
90% |
|
With
bromine; acetic acid;
|
|
|
With
nitric acid;
|
|
|
With
pyridine; bromine;
In
dichloromethane;
at 0 ℃;
|
|
|
With
pyridine; tert-butylhypochlorite;
In
dichloromethane;
at 0 ℃;
for 1h;
|
-
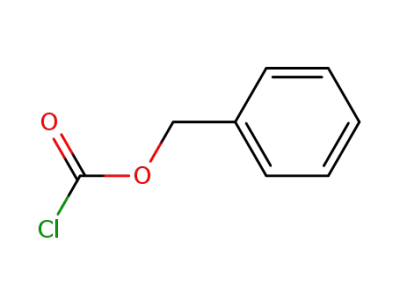
-
501-53-1,94274-21-2
benzyl chloroformate

-

-
2449-05-0
dibenzyl azodicarboxylate
| Conditions | Yield |
|---|---|
|
Multi-step reaction with 2 steps
1: N2H4
2: bromine; aqueous acetic acid
With
bromine; acetic acid; hydrazine;
|
|
|
Multi-step reaction with 2 steps
1: N2H4
2: concentrated HNO3
With
nitric acid; hydrazine;
|
|
|
Multi-step reaction with 2 steps
1: sodium carbonate; hydrazine / ethanol; water
2: pyridine; bromine / dichloromethane / 0 °C
With
pyridine; bromine; sodium carbonate; hydrazine;
In
ethanol; dichloromethane; water;
|
|
|
Multi-step reaction with 2 steps
1: hydrazine hydrate; pyridine / tetrahydrofuran / 0 °C
2: pyridine; tert-butylhypochlorite / dichloromethane / 1 h / 0 °C
With
pyridine; tert-butylhypochlorite; hydrazine hydrate;
In
tetrahydrofuran; dichloromethane;
|
2449-05-0 Upstream products
-
5394-50-3

bis(phenylmethyl) 1,2-hydrazinedicarboxylate
-
501-53-1

benzyl chloroformate
2449-05-0 Downstream products
-
37819-04-8
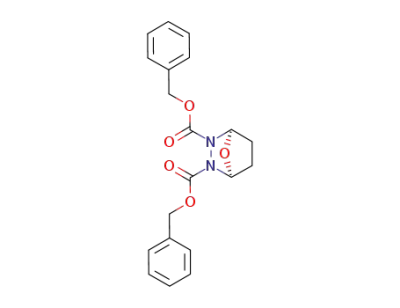
7-oxa-2,3-diaza-bicyclo[2.2.1]heptane-2,3-dicarboxylic acid dibenzyl ester
-
37819-02-6
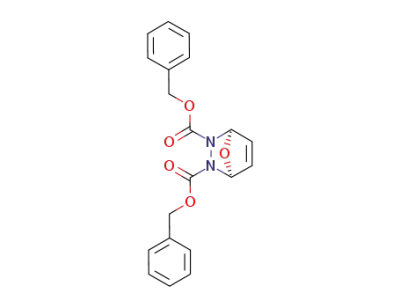
7-oxa-2,3-diaza-bicyclo[2.2.1]hept-5-ene-2,3-dicarboxylic acid dibenzyl ester
-
5321-35-7
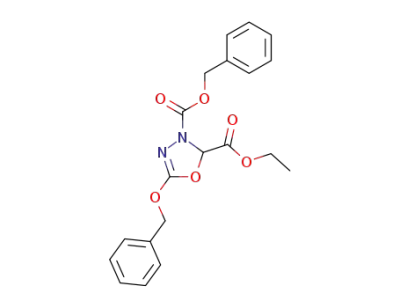
5-benzyloxy-[1,3,4]oxadiazole-2,3-dicarboxylic acid 3-benzyl ester 2-ethyl ester
-
5321-36-8
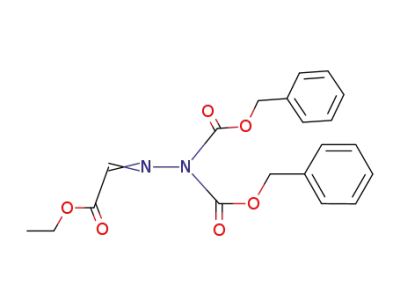
Glyoxylsaeureethylester-(N-dibenzyloxycarbonyl-hydrazon)
Relevant Products
-
2-Bromoacetamide
CAS:683-57-8
-
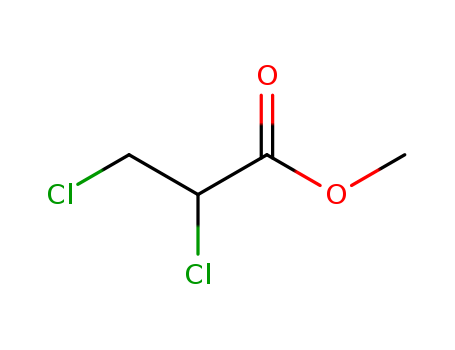
Methyl 2,3-dichloropropionate
CAS:3674-09-7
-
Methyl 3-cyclopentenecarboxylate
CAS:58101-60-3