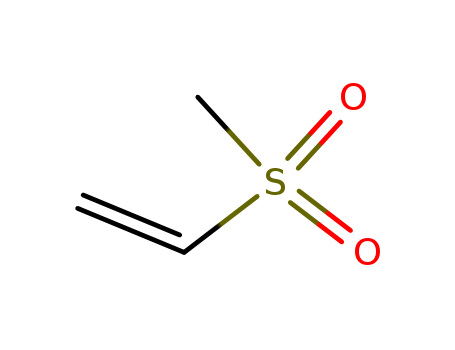
3680-02-2
- Product Name:Methyl vinyl sulfone
- Molecular Formula:C3H6 O2 S
- Purity:96% GC
- Molecular Weight:106.145
Product Details
Manufacturer supply Methyl vinyl sulfone 3680-02-2 with sufficient stock and high standard
- Molecular Formula:C3H6 O2 S
- Molecular Weight:106.145
- Appearance/Colour:light yellow liquid
- Vapor Pressure:0.0868mmHg at 25°C
- Refractive Index:n20/D 1.463(lit.)
- Boiling Point:115-117 ºC (19 mmHg)
- Flash Point:113 ºC
- PSA:42.52000
- Density:1.212
- LogP:1.25530
Methyl vinyl sulfone(Cas 3680-02-2) Usage
|
Air & Water Reactions |
Water soluble. |
|
Reactivity Profile |
Methyl vinyl sulfone is incompatible with oxidizing agents . Can react exothermically with reducing agents to release hydrogen gas or hydrogen sulfide. In the presence of various catalysts (such as acids) or initiatiators, can undergo exothermic polymerization reactions. |
|
Fire Hazard |
Methyl vinyl sulfone is probably combustible. |
|
Purification Methods |
Pass the sulfone through a column of alumina, then de-gas, distil it in a vacuum line and store it at -190o until required. [Beilstein 1 III 1866.] |
|
General Description |
Clear pale yellow liquid. |
InChI:InChI=1/C3H6O2S/c1-3-6(2,4)5/h3H,1H2,2H3
3680-02-2 Relevant articles
Process for preparing 2 - (methylsulfonyl) - ethylene oxide and derivatives thereof
-
Paragraph 0030-0032; 0043-0045, (2021/09/29)
The invention relates to 2 - (methylsulf...
Difluoro- and trifluoro diazoalkanes-complementary approaches in batch and flow and their application in cycloaddition reactions
Hock, Katharina J.,Mertens, Lucas,Metze, Friederike K.,Schmittmann, Clemens,Koenigs, Rene M.
supporting information, p. 905 - 909 (2017/08/14)
Herein we report on applications of fluo...
AZOLOPYRIDINE AND AZOLOPYRIMIDINE COMPOUNDS AND METHODS OF USE THEREOF
-
Page/Page column 129, (2012/03/26)
Provided herein are azolopyridine and az...
Episulfone substitution and ring-opening reactions via α-sulfonyl carbanion intermediates
Dishington, Allan P.,Douthwaite, Richard E.,Mortlock, Andrew,Muccioli, Adriano B.,Simpkins, Nigel S.
, p. 323 - 337 (2007/10/03)
Three-membered cyclic sulfones undergo s...
3680-02-2 Process route
-
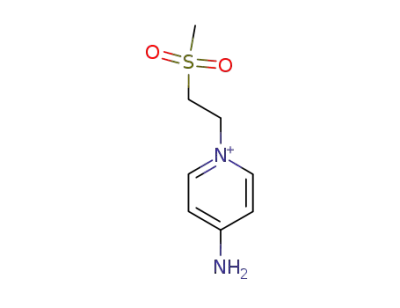
-
141375-65-7
4-Amino-1-(2-methanesulfonyl-ethyl)-pyridinium

-
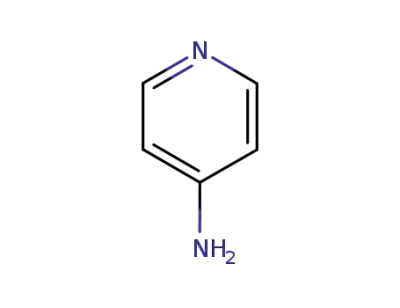
-
504-24-5
4-aminopyridine

-
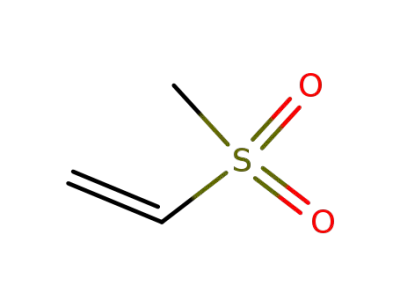
-
3680-02-2
methyl ethenyl sulphone
| Conditions | Yield |
|---|---|
|
With
water;
hydroxide;
at 25 ℃;
Equilibrium constant;
Mechanism;
|
-
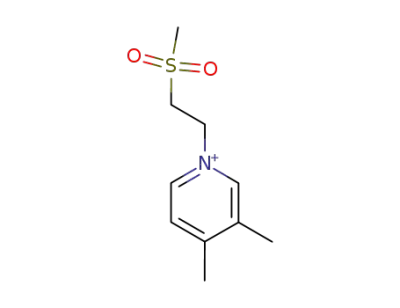
-
141375-69-1
1-(2-Methanesulfonyl-ethyl)-3,4-dimethyl-pyridinium

-
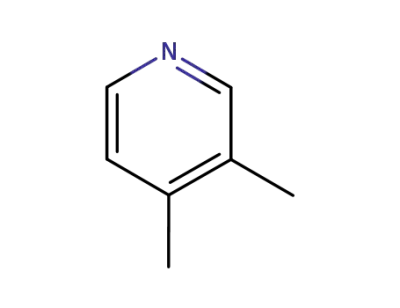
-
583-58-4
3,4-Lutidin

-

-
3680-02-2
methyl ethenyl sulphone
| Conditions | Yield |
|---|---|
|
With
water;
hydroxide;
at 25 ℃;
Equilibrium constant;
Mechanism;
|
3680-02-2 Upstream products
-
1822-74-8
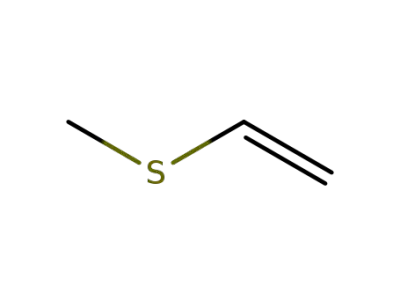
methyl vinyl sulfide
-
50890-51-2
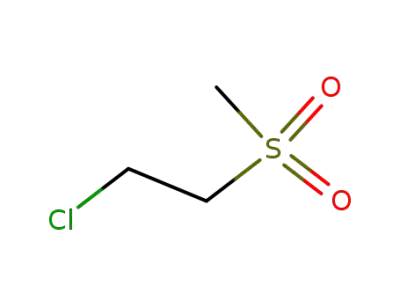
2-chloroethyl methyl sulfone
-
15205-66-0
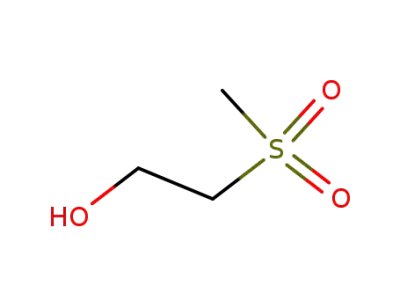
2-(methylsulfonyl)ethyl alcohol
-
92543-10-7
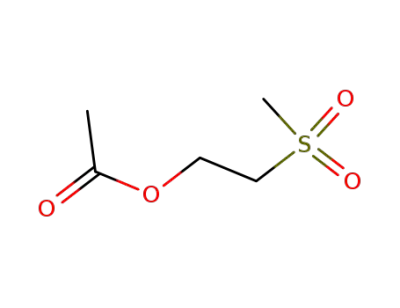
2-(methylsulfonyl)ethyl acetate
3680-02-2 Downstream products
-
153495-65-9
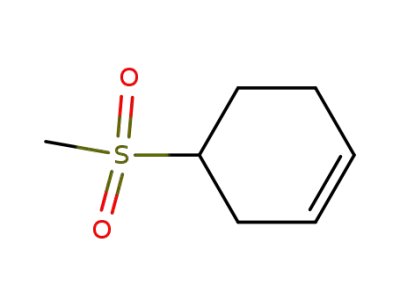
4-(methylsulfonyl)cyclohexene
-
5324-70-9
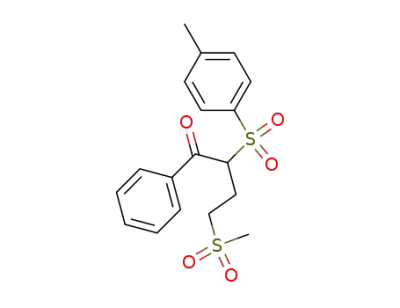
4-methanesulfonyl-1-phenyl-2-(toluene-4-sulfonyl)-butan-1-one
-
99987-26-5
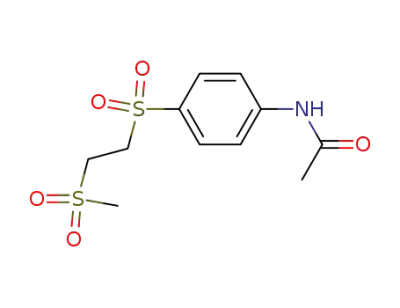
acetic acid-[4-(2-methanesulfonyl-ethanesulfonyl)-anilide]
-
5324-58-3
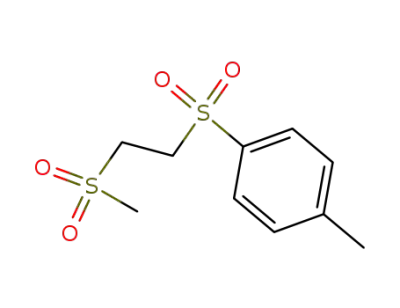
1-methanesulfonyl-2-(toluene-4-sulfonyl)-ethane
Relevant Products
-
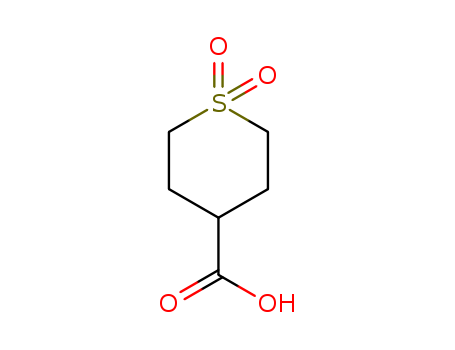
1,1-Dioxo-tetrahydrothiopyran-4-carboxylic acid
CAS:64096-87-3
-
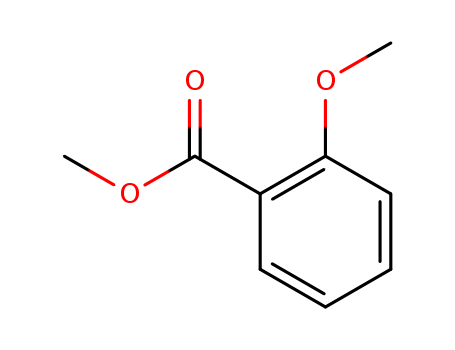
Methyl 2-methoxybenzoate
CAS:606-45-1
-
2-Phenyl-2-propanol
CAS:617-94-7